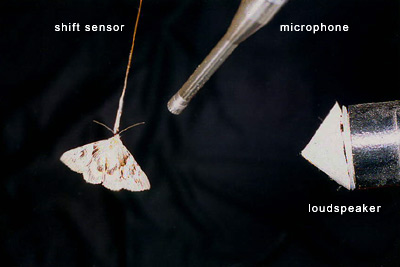
The retransmission method
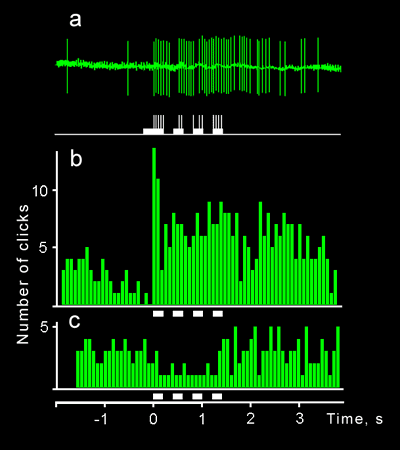
Before obtaining a proof of noctuids' ability to perceive the echoes of their own probing signals it was very important to determine which kind of behavior we should accept as a response to the echo. Earlier, similar problem arose and was solved in the course of studying responses to artificial stimuli in bats. It appeared that a good indicator of increased attention of a bat to biologically significant echolocation signals could be increased emission of the probing pulses (Grinnell, Griffin, 1958). Such changes were reproducible and could easily be registered.
Earlier we have found out that the maneuvres of a moth were very variable and often spontaneous (appearing without any noticeable stimulation). Moths had no certain sign of response to the echo: they demonstrated either movement to the object which was detected by means of echolocation or a detour manoeuvre, the changes of motivation occurring repeatedly during the experiment in some specimens. That was the other reason to search for a reliable behavioral test for moths.
The method we have finally elaborated was based on a principle of artificial echo retransmission: an echo from a non-existent (virtual) obstacle was retransmitted towards a moth after a certain delay in relation to it's own click. There was a dialog between the moth and the stimulating apparatus (some kind of self-stimulation). What is most important is that such stimulation system did not contain any movable details and thereby guaranteed that the results obtained were independent of the visual and tactile sensitivity of an insect. We have recorded the manouvres of a moth along with its clicks in every experiment but have not used them in further statistical analysis.
After the emission of each stimulating click, its repeated start up from the microphone was blocked during ca. 6 ms by means of a special electronic circuit. Thereby, the effects of direct action of the loudspeaker onto the microphone, and, as a consequence, the possibility to generate signals not related to the moth activity was eliminated.